
When mixed with water, acetic acid loses a positively charged ion (H+).

It has the chemical formula CH 3COOH (or HC 3H 2O 2). The reaction that occurs is an example of acid-base chemistry. Add the second solution to the first, stand back and watch what happens. The other is a mix of baking soda and water. One solution contains vinegar, dish soap, water and a little food coloring. Explainer: What are acids and bases?Ī baking soda volcano’s foamy rush is the result of a chemical reaction between two solutions. All that’s needed is a hypothesis to test - and more than one volcano. The whole thing looks like it was put together the morning of the fair.īut it’s not too difficult to turn this easy science demo into a science experiment. That clay mountain “smoking” in front of a poster board can be kind of sad, though. It’s a science fair staple: the baking soda volcano. You can repeat the steps here and compare your results - or use this as inspiration to design your own experiment.
#How to make baking powder chemical formula series
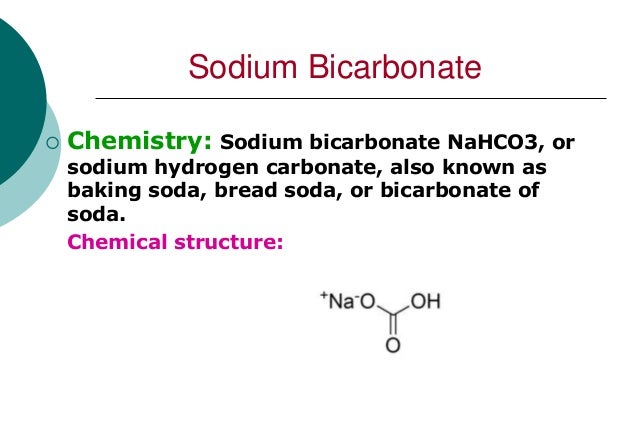
Each oxygen atom in the chemical formula is made up of 8 protons, 8 electrons, and 8 neutrons with a number of 2 energy levels (Bentor, n.d.).This article is one of a series of Experiments meant to teach students about how science is done, from generating a hypothesis and designing an experiment to analyzing the results with statistics. The carbon atom is made up of 6 electrons, 6 protons, and 6 neutrons with a number of 2 energy levels. The hydrogen atom is made up of one proton, one electron, and no neutrons with only 1 energy level. The sodium atom is made up of 11 electrons, 11 protons, and 12 neutrons with a number of 3 energy levels. This chemical compound is made up of 1 sodium atom, 1 hydrogen atom, 1 carbon atom, and 3 oxygen atoms (Sodium Bicarbonate, 2006). The chemical formula for baking powder is NaHCO3. This happens because when sodium bicarbonate is dissolved or heated in water, it produces carbon dioxide gas (CO 2 ).This item is also known as sodium bicarbonate or sodium hydrogen carbonate .
A leavening agent is a substance that causes dough or batter to rise. Since there were a lot of bubbles foaming, there was a lot of carbon dioxide produced.The reaction between the hot water and the baking powder was very strong compared to the cold water and baking powder mixed together (Sodium Bicarbonate, 2006).īaking powder is well known to be used in baked goods as a leavening agent. This is because the heat from the water made the baking powder react more than the cold water. If hot water is mixed in with baking powder, it would have a major affect to the mixture. This means that carbon dioxide was present during the reaction. When the cold water and the baking powder was mixed together, there were only little bubbles that were produced. If baking powder was mixed with cold water, the mixture would be affected very little. When acids are present in the mixture, they produce carbon dioxide gas (Romanowski, 2006). When baking powder is mixed with water, it produces sodium ions and bicarbonate ions. Sodium bicarbonate has a melting point of about 50 degrees celsius (Sodium Bicarbonate, 2006). It is an odourless, white, powdery substance. The picture above is a household item called "baking powder".
